The Science of Creatine Monohydrate: A Narrative Review of Mechanisms, Efficacy and Safety
An evidence synthesis spanning endogenous biosynthesis, pharmacokinetics, ergogenic performance, neurological applications, and clinical safety profile, drawn from researched and peer-reviewed sources.
The Science Hub Editorial Group
Editorial review by experts in sports nutrition, exercise physiology, and clinical pharmacology.
Abstract
Background
Creatine, a guanidino compound endogenously synthesized in the kidney and liver and obtained dietarily from animal-source foods, has been studied as an ergogenic and therapeutic agent for over three decades. Despite a substantial evidence base, public-facing claims and supplement marketing continue to diverge from the peer-reviewed literature.
Objective
To provide a neutral, source-attributed synthesis of the current evidence on creatine monohydrate, addressing biochemistry, pharmacokinetics, ergogenic performance, neurological applications, women's-health applications, dosing, and safety, and to make explicit where evidence is robust, where it is preliminary, and where it is contested.
Sources
Multiple peer-reviewed sources were selected, prioritizing systematic reviews and meta-analyses (n = 14), position stands from recognised scientific bodies (n = 3), randomized controlled trials (n = 23), and mechanistic studies (n = 18). Searches covered PubMed, PMC, and recent Frontiers and JISSN issues through May 2026.
Principal Findings
Commercial creatine monohydrate is produced through a four-step synthetic process — condensation, crystallisation, purification, and milling — with post-crystallisation milling determining particle grade: standard (100–500 µm), micronized (10–100 µm), ultra-micronized (1–10 µm), and nano-grade (below 1 µm). Smaller particles dissolve faster, but thermodynamic solubility remains fixed at 13 g/L regardless of particle size. Once dissolved, intestinal absorption via the CRT1 transporter is near-complete across all grades, and muscle creatine accumulation plateaus at approximately 150–160 mmol/kg dry mass irrespective of particle grade. Micronization improves dissolution speed, mouthfeel, and formulation flexibility — not ergogenic outcomes.
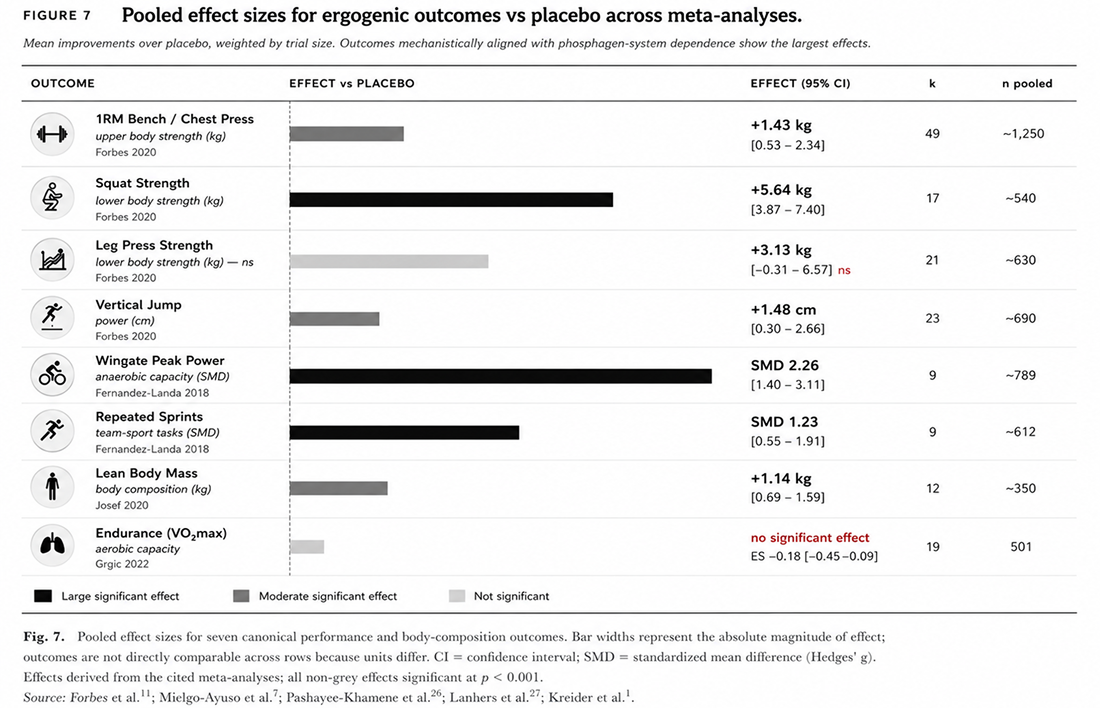
Physiologically, supplementation elevates muscle phosphocreatine stores by 15–40% at maintenance doses of 3–5 g/day, directly enhancing ATP resynthesis during high-intensity efforts. Pooled meta-analytic data confirm consistent improvements in maximal strength, anaerobic power, and lean body mass alongside resistance training. No significant adverse effects have been identified at doses up to 30 g/day over five years.
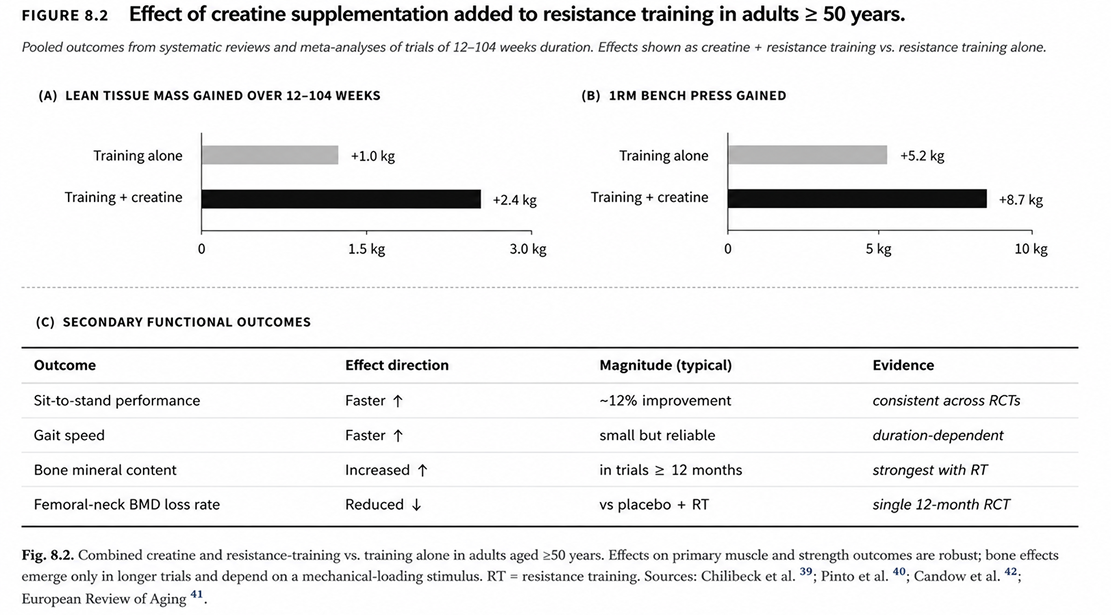
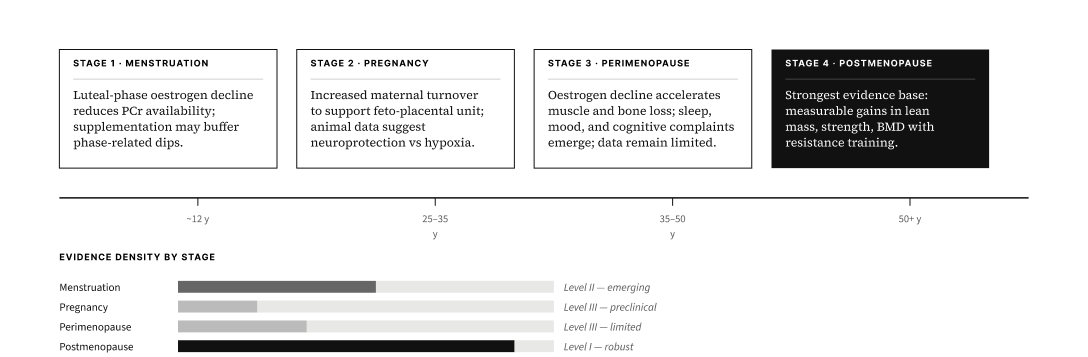
Emerging evidence extends applications to cognition under metabolic stress, mood disorders, traumatic brain injury recovery, female-specific physiology, and sarcopenia — a growing body of research underpinned by creatine's established role in neuronal energy homeostasis.
Limitations
Several recent cognition meta-analyses contain unit-of-analysis errors that may have inflated reported effect sizes. Long-term data in pregnancy, paediatric populations, and pre-existing kidney disease remain limited. Most trials are of short-to-medium duration relative to typical lifetime use patterns.
Contents
Chemical identity and tissue distribution
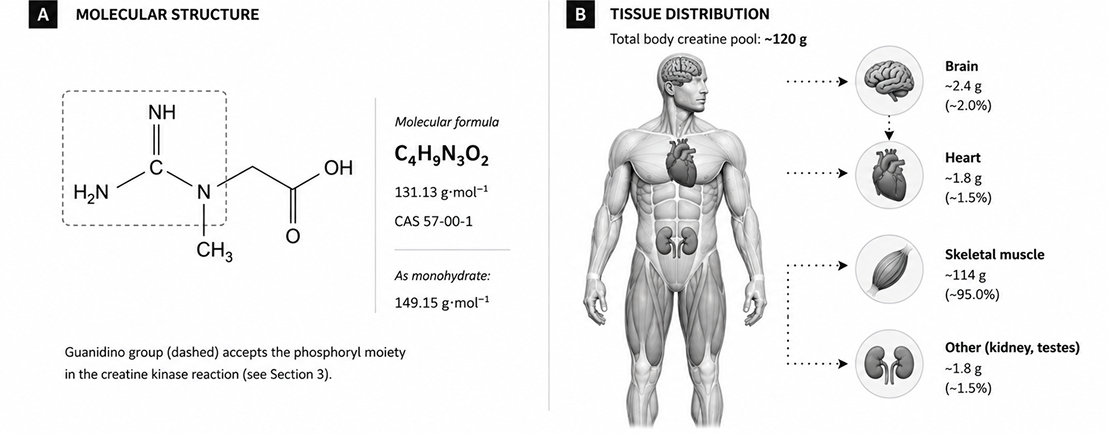
Creatine (α-methylguanidinoacetic acid; C4H9N3O2) is a non-protein nitrogenous compound of the guanidine phosphagen family. Its molecular mass is 131.13 g/mol; in its most stable crystalline solid it carries a single co-crystallized water molecule, yielding creatine monohydrate (149.15 g/mol) with theoretical water content of 12.07% w/w [1,2].
Across a 70 kg adult, the total body pool is approximately 120 g of creatine, of which around 95% resides in skeletal muscle as a dynamic equilibrium between free creatine and phosphocreatine. The remaining ~5% is distributed across the brain, myocardium, kidneys, and testes [3,4]. Within muscle tissue, approximately two-thirds of the creatine pool is phosphorylated (phosphocreatine, PCr) under resting conditions; the ratio shifts rapidly toward free creatine during high-intensity contraction.
The molecule’s most physiologically consequential structural feature is the guanidino group at C-2, which serves as the phosphoryl acceptor in the creatine kinase reaction. This single chemical detail – a transferable high-energy phosphate bond on a small, water-soluble molecule – is the basis for creatine’s role in cellular energy buffering.
Baseline muscle creatine concentration varies by diet. Omnivores, who obtain 1–2 g/day of creatine from meat and fish, exhibit muscle total-creatine values of approximately 120 mmol/kg of dry muscle. Vegetarians and vegans, who rely entirely on endogenous synthesis, exhibit baseline values approximately 10–30% lower [1,19]. This baseline difference is associated with a correspondingly larger absolute response to supplementation in plant-based individuals – a point returned to in Section 7.
Daily creatine turnover in adults is approximately 1.7% of the total body pool, corresponding to roughly 2 g of creatine that is converted irreversibly to creatinine (a spontaneous cyclisation product) and excreted in urine. This loss must be balanced by some combination of endogenous synthesis and dietary intake, the proportions of which vary by individual [3,4].
A two-enzyme inter-organ pathway from three amino acids
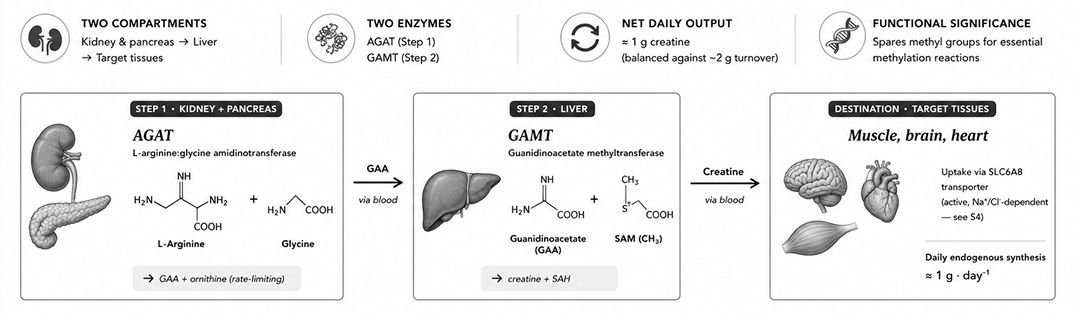
Creatine synthesis requires three amino acids, glycine, arginine and methionine, and three enzymes, methionine adenosyltransferase (MAT), arginine:glycine amidinotransferase (AGAT) and guanidinoacetate methyltransferase (GAMT) [3,5].
The rate-limiting first reaction is catalysed by L-arginine:glycine amidinotransferase (AGAT; EC 2.1.4.1), with highest activity in renal cortex and pancreas whereas the activity of GAMT is highest in the liver. AGAT transfers a guanidino group from arginine to glycine, yielding guanidinoacetic acid (GAA) and ornithine. GAA enters circulation and is taken up by hepatocytes, where the second enzyme, guanidinoacetate methyltransferase (GAMT; EC 2.1.1.2), transfers a methyl group from S-adenosylmethionine (SAM) to yield creatine and S-adenosylhomocysteine. Newly synthesized creatine returns to circulation and is taken up actively by extra-hepatic tissues via the SLC6A8 transporter [3,4].
AGAT is down-regulated by intracellular creatine, providing the principal regulatory mechanism. This explains why supplementation effectively replaces — rather than adds to — endogenous synthesis in the short term, sparing the methylation cost detailed below [6].
The methylation step of creatine synthesis imposes a considerable burden on methyl group balance and methionine metabolism, as it consumes approximately 40% of all S-adenosylmethionine (SAM) utilized by methyltransferase reactions in the body. SAM is regenerated endogenously through methylneogenesis, a process critically dependent on B-vitamin status. This pathway requires the sequential actions of three enzymes: serine hydroxymethyltransferase (SHMT), which generates one-carbon units from serine; 5,10-methylenetetrahydrofolate reductase (MTHFR), which catalyzes the irreversible reduction of 5,10-methylenetetrahydrofolate to 5-methyltetrahydrofolate using NADPH (derived from niacin) as a reductant and FAD (derived from riboflavin) as a prosthetic group; and methionine synthase (MS), which transfers the methyl group from 5-methyltetrahydrofolate to homocysteine to regenerate methionine. Notably, MS is one of only two known mammalian enzymes that require vitamin B12, in the form of methylcobalamin, as a cofactor. The dependence of this regenerative cycle on folate, riboflavin, niacin, and cobalamin underscores the extent to which creatine synthesis places a significant demand on one-carbon metabolism and overall methyl group homeostasis [5].
Inborn errors of this pathway — AGAT deficiency, GAMT deficiency, and SLC6A8 transporter deficiency — collectively define a recognised class of creatine deficiency disorders presenting with intellectual disability, speech-language impairment, and epilepsy. Oral creatine supplementation partially corrects the first two but is largely ineffective in transporter deficiency, in which the gateway into target tissues is itself defective [6].
Bioenergetic role of phosphocreatine and the creatine kinase reaction
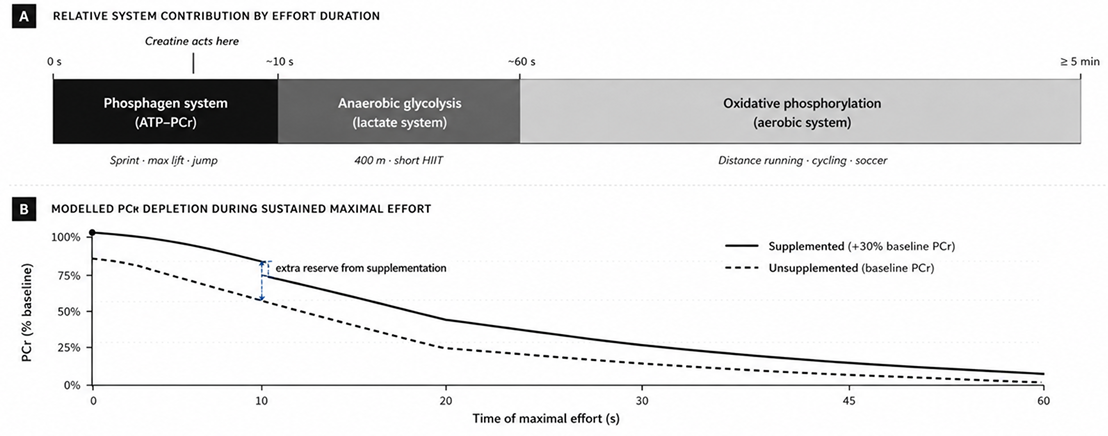
Adenosine triphosphate (ATP) is the proximate energy currency of muscle contraction, yet skeletal muscle stores sufficient ATP for only approximately two seconds of maximal effort. Three sequential bioenergetic systems sustain longer activity: the phosphagen (ATP-phosphocreatine) system, anaerobic glycolysis, and oxidative phosphorylation. These systems overlap continuously rather than switching discretely; their relative contribution depends on the duration and intensity of work.
The creatine kinase reaction
Creatine kinase (CK; EC 2.7.3.2) catalyses a single readily reversible reaction that runs near equilibrium in cytoplasm and mitochondria:
Because the reaction operates near equilibrium and CK has exceptionally high catalytic activity, ATP can be regenerated within milliseconds of being hydrolysed during muscle contraction. During recovery, ATP produced by oxidative phosphorylation runs the reaction in reverse to rephosphorylate creatine.
A widely accepted refinement of this model — the phosphocreatine shuttle hypothesis — proposes that phosphocreatine, not ATP, is the principal carrier of high-energy phosphate through cytoplasm. PCr is smaller, electrically neutral, and diffuses faster than ATP, allowing efficient energy transfer from mitochondrial CK isoforms to the myofibrillar CK pool adjacent to the contractile apparatus [6].
Effect of supplementation on PCr stores
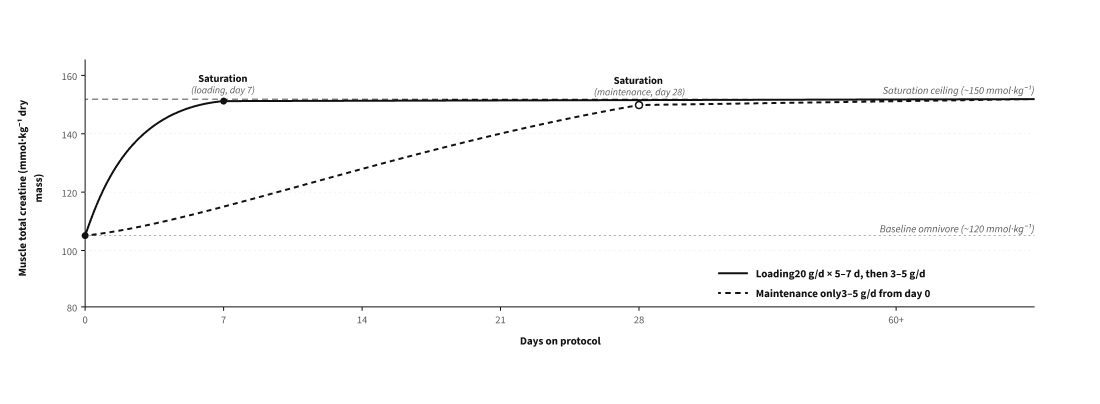
Oral creatine supplementation at 20 g/day for 5–7 days, or 3–5 g/day for 3–4 weeks, increases muscle total creatine and phosphocreatine content by approximately 15–40% above baseline in most individuals [1,8,9]. Magnitude of response correlates inversely with baseline content: individuals with low baseline (vegetarians; older adults) gain more, and individuals already at or near saturation (~150 mmol/kg dry muscle) gain little.
The functional consequence of elevated PCr stores is a delayed onset of the phosphagen-system-limited fatigue, observable as more repetitions tolerated at submaximal loads, greater work output across repeated sprints, and a higher anaerobic-work ceiling before glycolytic and oxidative systems must contribute. Section 7 presents the pooled meta-analytic evidence on these outcomes.
(b) Modelled PCr depletion during sustained maximal effort, with and without prior creatine supplementation.
"Phosphocreatine functions not only as an immediate ATP-regenerating buffer, but also as the principal molecular vehicle by which high-energy phosphate is transferred from mitochondrial sites of production to the myofibrillar sites of consumption."
— Wallimann, Wyss, Brdiczka, Nicolay & Eppenberger, Biochemical Journal, 1992
Stages of disposition: dissolution, absorption, distribution, and tissue uptake
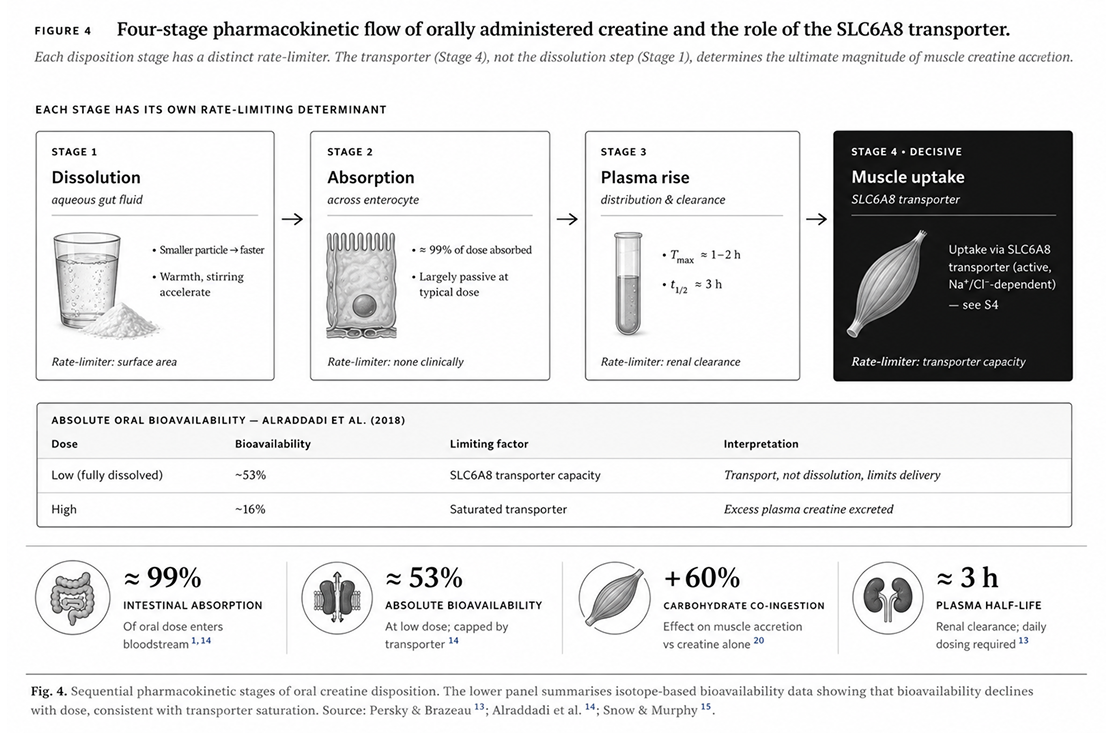
Orally administered creatine traverses four sequential disposition stages, each with its own kinetic determinants. Conflation of these stages — particularly equating dissolution speed with muscle uptake — underlies much of the marketing differentiation among commercial creatine forms [13,14].
Dissolution occurs in gastric and intestinal fluid; it is faster with smaller particles, warmer temperatures, and active stirring. At typical doses (3–5 g in 250 mL water), standard-grade monohydrate dissolves within approximately two minutes; micronized grades within approximately one to two minutes. Intestinal absorption is essentially complete: stable-isotope studies indicate that approximately 99% of an oral creatine dose enters the bloodstream, with the small remainder accounting for unabsorbed compound passing into the colon [14].
Plasma concentrations peak at approximately 1–2 hours post-ingestion, with an elimination half-life of approximately three hours via renal clearance. The decisive stage is the fourth: transit from plasma into target tissue via the SLC6A8 creatine transporter (also termed CreaT or CrT1).
SLC6A8: a saturable active transporter
SLC6A8 is a member of the SLC6 sodium- and chloride-dependent neurotransmitter transporter family. It moves creatine against its concentration gradient at a stoichiometry of 2 Na+ : 1 Cl- : 1 creatine.
Expression is highest in skeletal muscle and heart, with substantial expression also in brain, kidney, colon, and testes [14,15].
Three features of this transporter govern the pharmacokinetics of supplementation:
First, SLC6A8 is saturable. Above its Vmax, additional plasma creatine cannot be translocated faster; the excess is filtered by the glomerulus and excreted in urine. This ceiling explains why doses substantially exceeding 5 g/day produce diminishing returns and why isotope studies show absolute oral bioavailability falling from ~53% at low dose to ~16% at high dose [14].
Second, SLC6A8 activity is upregulated by insulin via the SGK1/PIKfyve signalling axis, which increases membrane trafficking of the transporter. Co-ingestion of creatine with approximately 50–100 g of carbohydrate has been shown to increase muscle accretion by ~60% relative to creatine alone — a finding originally demonstrated by Green et al. using direct muscle biopsy [17,21].
Third, SLC6A8 is the only physiological entry route into skeletal muscle and brain cells for creatine. Knockout studies in mice confirm that, in its absence, muscle creatine content falls to ~18% of wild-type values despite the presence of biosynthetic enzymes [18]. Loss-of-function variants in humans cause SLC6A8 deficiency, a form of cerebral creatine deficiency that is largely unresponsive to oral creatine therapy.
The practical implication of transporter-limited uptake is that further increases in dissolution speed — through micronization, chemical salt forms, or co-formulation with surfactants — produce no additional muscle creatine accretion once the small dose is fully dissolved. This argument is examined in Section 5, where head-to-head trials of alternative commercial forms are evaluated against this mechanistic prediction.
Comparative evaluation of creatine forms in head-to-head trials
Following the commercial success of creatine monohydrate (CrM), supplement manufacturers introduced multiple alternative forms — esters, salts, chelates, and pH-buffered formulations — accompanied by claims of superior absorption, smaller required doses, reduced water retention, or improved gastrointestinal tolerance. Most such claims have been evaluated in randomized controlled trials and three successive position papers (Jäger et al. 2011, Kreider et al. 2017, Antonio et al. 2021).
Across these evaluations, no alternative form has shown superior performance outcomes to monohydrate in head-to-head trials, and several have been shown to be inferior.
Table 1 summarises the current state of the evidence for the nine commercial forms most frequently encountered. Evidence-strength designations reflect the volume and quality of head-to-head comparisons available; "limited" indicates that human RCT data are insufficient to draw conclusions, not that the form is necessarily ineffective.
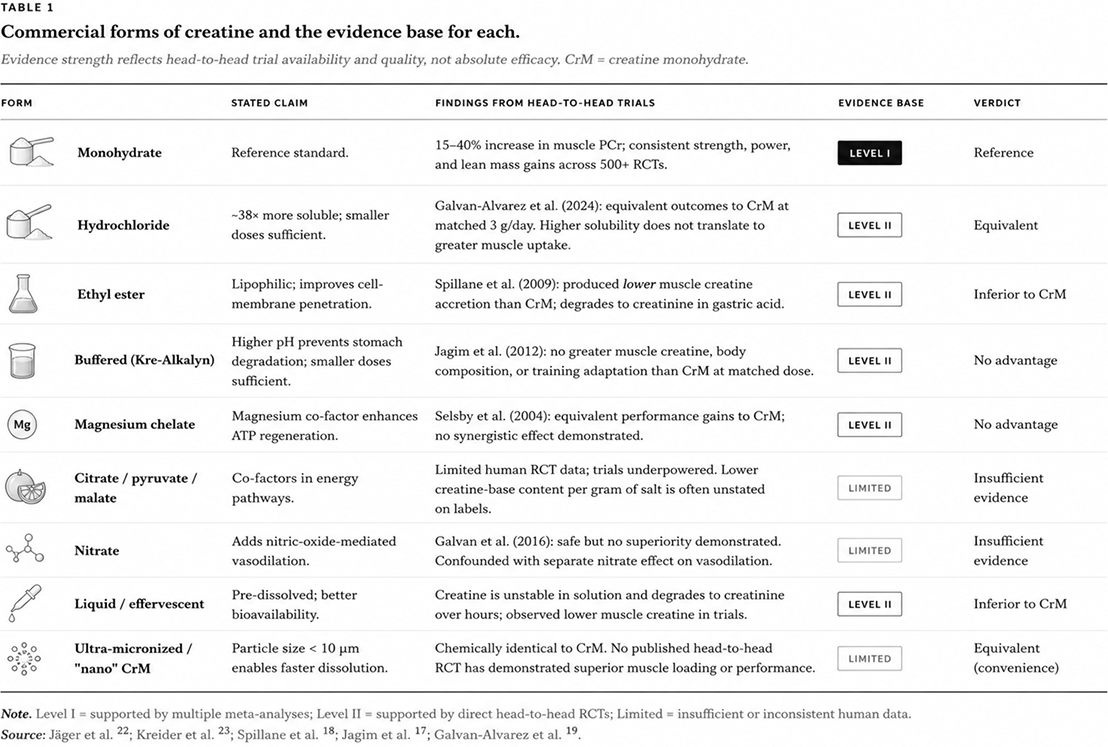
Two of these forms warrant individual attention because of their popularity or the persistence of marketing claims relative to the published evidence.
Creatine ethyl ester (CEE) was introduced on the rationale that esterification would improve lipophilicity and therefore membrane penetration. The Spillane et al. (2009) double-blind RCT in 30 resistance-trained men directly tested this hypothesis: participants received CEE, CrM, or placebo for 47 days alongside structured resistance training. The CEE group exhibited lower serum creatine and lower intramuscular creatine accretion than the CrM group, alongside markedly elevated serum creatinine consistent with rapid degradation of CEE to creatinine in the gastrointestinal tract [17]. This finding has not been overturned in subsequent trials.
Creatine hydrochloride (Cr-HCl) has more nuanced evidence. Its solubility advantage is real — laboratory measurements indicate solubility approximately 38-fold higher than monohydrate — but the 2024 trial by Galvan-Alvarez et al. in elite handball and softball players compared low-dose CrM (3 g/day) with low-dose Cr-HCl (3 g/day) and found equivalent improvements in neuromuscular performance and strength, with the conclusion that "claims of Cr-HCl superiority are unfounded and misleading" [19]. The mechanism-derived prediction — that solubility differences do not propagate to muscle uptake once dose is fully dissolved — was confirmed.
Manufacturing route and the science of particle-size spectrum
6.1 Raw materials and core chemistry
The dominant industrial production route for creatine monohydrate has remained essentially unchanged for three decades. Sarcosine (N-methylglycine) and cyanamide (H₂N-C≡N) react in aqueous solution via nucleophilic addition at 90–100 °C, pH 7–9, for 2–6 hours. Sarcosine supplies the methylated nitrogen backbone; cyanamide donates the guanidino group. Purified deionised water is used throughout. Impurity controls target dicyandiamide (DCD ≤50 ppm), residual cyanamide, and heavy metals.59,60
The critical process-related impurities are DCD — an unreacted precursor arising from incomplete cyanamide dimerization — and dihydrotriazine (DHT), a cyclization by-product formed preferentially at elevated temperature or alkaline pH during crystallization. Standard industrial processes yield DCD at 30–80 ppm; an integrated ultra-fine process achieves ≤15 ppm without additional downstream purification by maintaining near-neutral pH (7.0–7.5) and a stoichiometric excess of sarcosinate (1.05–1.10 mol equiv.), which drives cyanamide to near-complete consumption. DHT is held below 1 ppm by controlling crystallization temperature below 85 °C.59,60
6.2 Step-by-step manufacturing process
Table 2 summarises the ten-stage production sequence from precursor preparation through quality-control release. Critical control parameters at each stage determine both purity and particle characteristics of the final product. The Creapure® benchmark — ≥99.99% assay, DCD <10 ppm, DHT not detected, heavy metals <5 ppm — represents the highest publicly documented purity standard for commercial creatine monohydrate.59

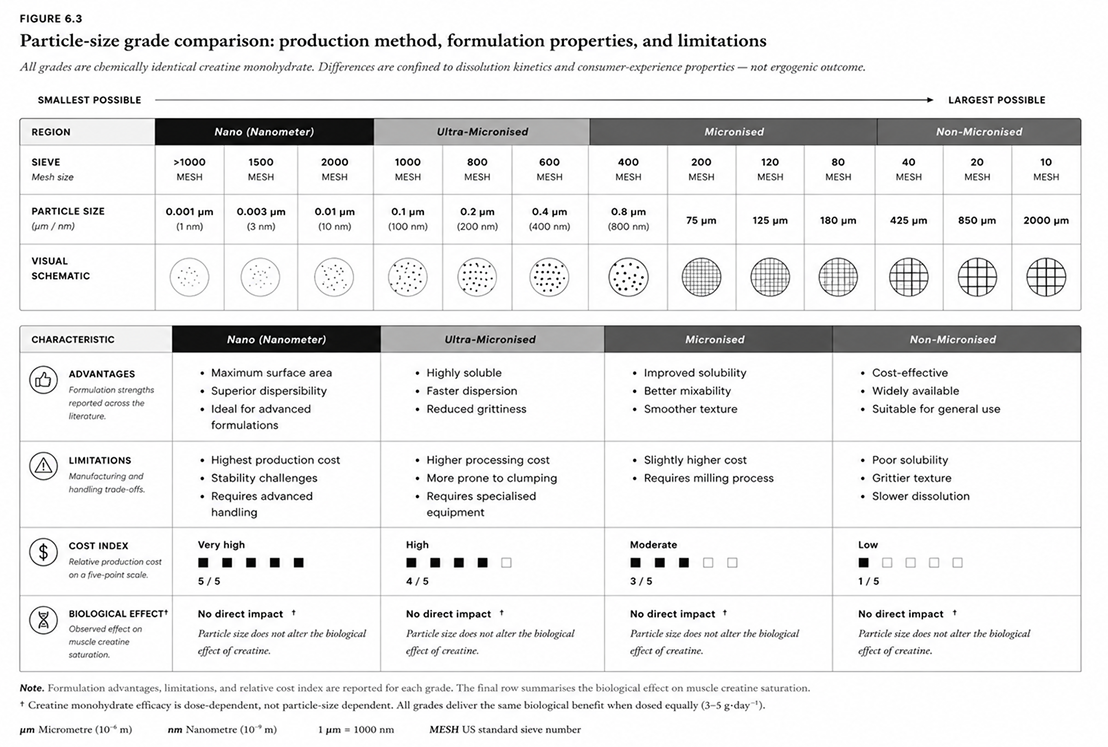
6.3 Particle size progression — standard to nano
Primary milling yields standard CrM. Further processing reduces particle diameter through distinct technology tiers, each with specific production methods, advantages, and practical limitations. The dissolution-rate difference across grades is real and reproducible. A 5 g serving in 250 mL of unstirred water at 25 °C dissolves to 95% completion in approximately 288 seconds (4.8 min) for standard grade, 142 seconds for micronized, and 38 seconds for ultra-fine — an eight-fold range. Reducing particle size from ~180 µm to ~6 µm increases the geometric surface-area-to-volume ratio approximately 30-fold, driving this difference in accordance with the Noyes–Whitney equation.59,60
Ultra-fine production requires air-jet milling under nitrogen purge at ≤30% relative humidity to prevent moisture-induced agglomeration; the milling zone is held below 45 °C, above which the monohydrate converts to the anhydrous form. Nano-grade material (<1 µm) can be produced by wet nanomilling, antisolvent precipitation, or amorphous co-grinding, but carries the highest physical instability — requiring polymer stabilisers and presenting Ostwald ripening risk in aqueous suspension. No published RCT has demonstrated superior muscle creatine tissue saturation for nano-grade vs. standard CrM at equivalent doses.59

6.4 The pharmacokinetic cascade: particle size → tissue creatine

6.4.1 Solubility — Thermodynamic Ceiling
Creatine monohydrate solubility in water at 25 °C: ~13 g/L (0.013 g/mL). A standard 5 g dose requires ~385 mL to dissolve completely at room temperature.1 Solubility is a fixed thermodynamic property — particle-size reduction does NOT change it. Chemical modification (e.g., creatine HCl: ~679 mg/mL ≈ 52× higher) requires altering the molecule, formally yielding a new chemical entity.59
Apparent (kinetic) solubility: Amorphous and nano-grade CM can produce transient supersaturation above Cs through elevated surface free energy (Ostwald–Freundlich effect) or disordered lattice energy.59,69 Patents report 3–10× kinetic solubility enhancement, but these states are metastable and revert to the thermodynamic limit at equilibrium.69,70
6.4.2 Dissolution — The Kinetic Variable Particle Size Controls
Dissolution rate is the primary — and essentially only — pharmacokinetic variable responsive to particle size. As described by the Noyes–Whitney equation (dM/dt = D·A·Cs/h × (Cs − Ct)), reducing particle diameter by 10× increases specific surface area by ~10×, proportionally accelerating dissolution rate. This is the primary — and essentially only — pharmacokinetic variable responsive to particle size.59,72